Health
COVID-19 vaccinations for kids under 5 could start June 21
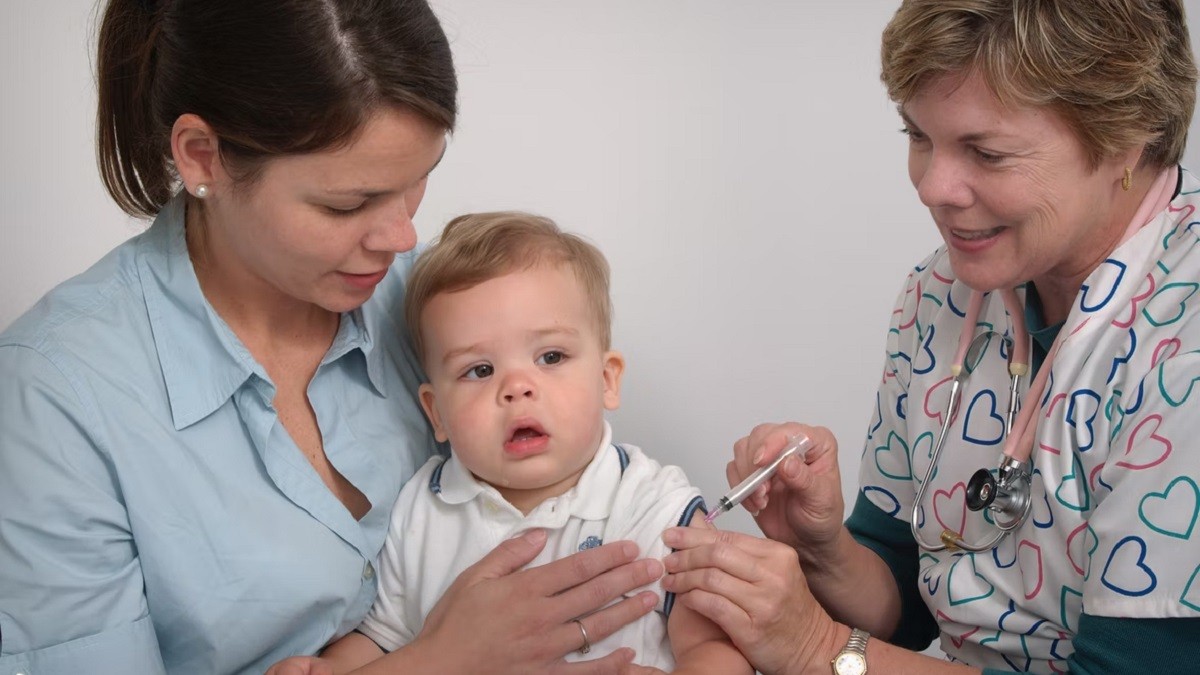
- Kids under 5 could receive COVID-19 vaccinations as early as June 21, the White House said.
- The schedule would still depend on authorization from the FDA.
- Pfizer said last month that its vaccine for young children was about 80 percent effective after the third shot.
COVID-19 vaccinations for kids under 5 could start as early as June 21, pending authorization from the Food and Drug Administration (FDA), the White House confirmed on Thursday.
White House COVID-19 response coordinator Ashish Jha stated, “We expect vaccinations will begin in earnest as early as Tuesday, June 21, and really roll on throughout that week.”
The applications from Pfizer and Moderna are set to be discussed by the FDA advisory committee on June 14 and 15, after which a decision on authorization can be expected “soon thereafter,” Jha said.
A sign-off would also be needed from the Centers for Disease Control and Prevention (CDC).
Some parents have been frustrated by the delays since kids under 5 are the only age group that still has no available vaccine.
Jha acknowledged that it will take some time to kickstart the vaccination program, but he’s optimistic that “within weeks every parent who wants their child to get vaccinated will be able to get an appointment.”
It’s unknown how many parents are willing to get their young children vaccinated, however. Some have expressed eagerness to do so as soon as a vaccine is available, but vaccination rates have lagged for older children. The CDC reported only about 30 percent vaccination rates for kids aged 5 to 11.
Last month, Pfizer released data that showed its vaccine for young children was about 80 percent effective after the third shot. They have previously delayed their application while waiting for results from a third dose.
There will be “plenty of supply” of the vaccine for young children, Jha said, with about 10 million doses to be initially released to states, pharmacies, and community health centers.
Vaccine orders can be placed starting on Friday, but the shipping of doses cannot start until authorization from the FDA.
Source: The Hill

Eileen Ross
June 3, 2022 at 7:27 pm
When are they going to start arresting these poison jabbing monsters???????
Sean Richman
June 3, 2022 at 8:23 pm
Be very careful what you wish for.My wife and I are in our eighties,and I agree that our age matters,but we have lost so much memory,concentration,energy and attitude with only two phizer pricks that we would never,ever do it again.We did what our doctors told us to do,which is worse mental decline or the domocRATS/chinese so called covid.
Lyudmila
June 3, 2022 at 11:26 pm
Moms, take care of your children, Fauci don’t need them.
Notolerance
June 8, 2022 at 7:53 am
Before there was ever a vaccine, way over ninety percent of the people in this country that got covid had almost unnoticeable symptoms. Loss of taste, slight headache, mild fever. Then, the pharmaceutical companies developed the vaccine and claimed it was over ninety percent effective to reduce symptoms. They used a statistic that naturally existed without the vaccine. They are still using statistics that naturally exist without the vaccine! They are getting filthy rich sucking up the taxpayers’ money, injecting an ineffective substance into people’s bodies. The fact is, their vaccine doesn’t work, and it causes nasty side effects in many people. The fact is, children in general have no issues with covid, so why would any parents be fooled by the Biden Administration. Don’t let them poison your children!